Regulatory complexity that takes years to navigate without the right expertise
India market expansion
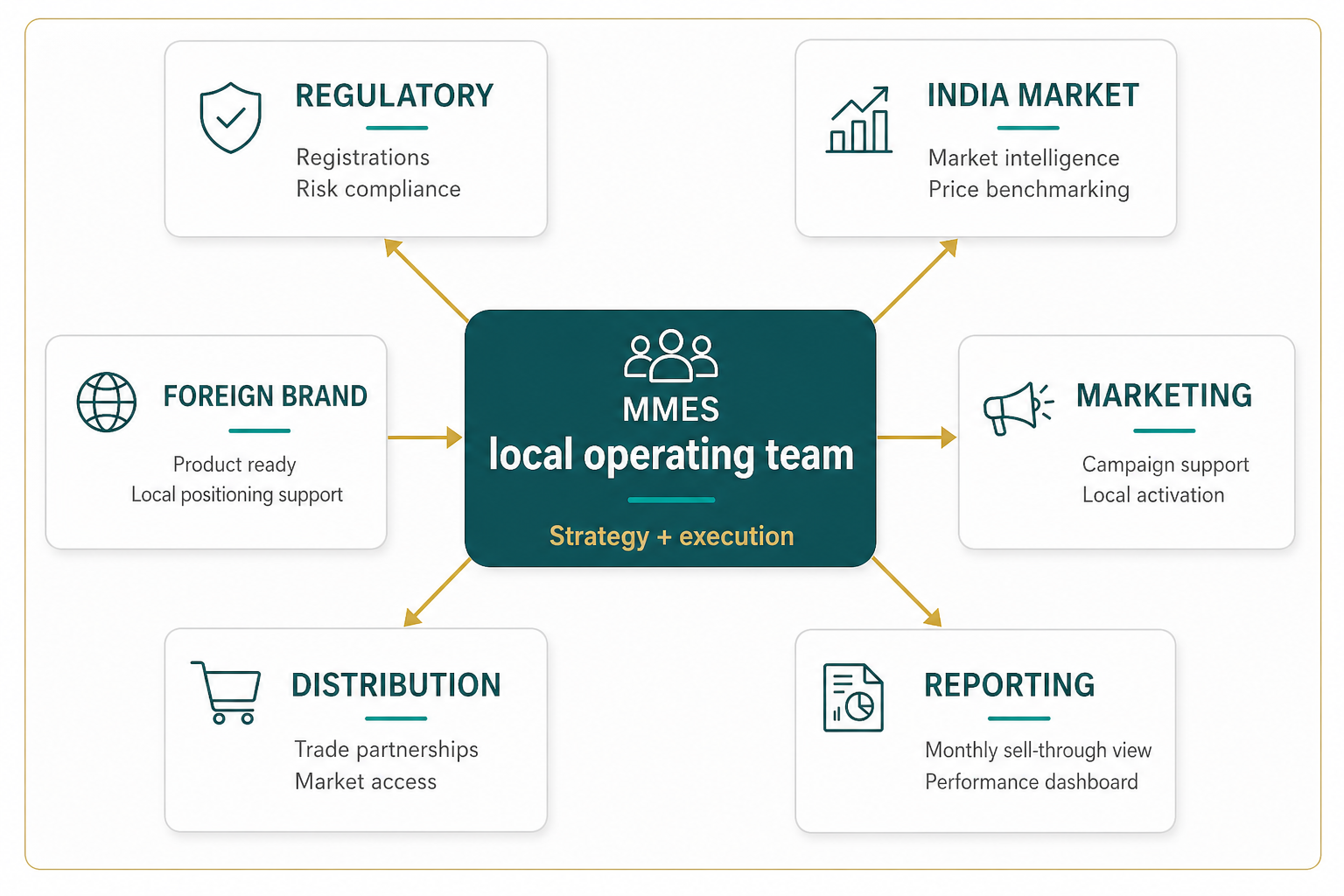
Your commercial team in India. Without the overhead.
MMES helps foreign brands enter India, handling regulatory approvals, import operations, distribution, and sales, so you build revenue, not bureaucracy.
The challenge
India is one of the world's biggest opportunities. It's also one of the hardest to enter.
Distribution networks built on relationships you haven't had time to build
The fixed cost of a local team that kills your margins before you've found your first customer
We solve all three.
Our services
One partner. Every commercial function.
Market Entry Strategy
Understand the opportunity before committing capital.
Learn more ->Regulatory & Compliance
Navigate FSSAI, CDSCO, BIS, and Customs without the confusion.
Learn more ->Import & Trade Operations
From port to warehouse, managed end to end.
Learn more ->Sales & Distribution
A sales team that already knows the market.
Learn more ->Marketing Support
Positioning that resonates in the Indian market.
Learn more ->Financial Operations
Invoicing, collections, and reconciliation, handled.
Learn more ->How it works
From signed agreement to first sale, we run the operation.
Step 1 - Assess
We evaluate your product, your category, and the regulatory pathway. You get a clear market entry plan.
Step 2 - Establish
We file registrations, build your distributor network, and set up your import operations.
Step 3 - Grow
We run sales, manage distributor relationships, and report sell-through monthly. You scale.
Who we serve
Sector expertise that matters in a regulated market.
Nutraceuticals & Wellness
India's nutraceutical market is growing quickly. We navigate FSSAI product approvals, build pharmacy and health retailer networks, and position your brand in a category where Indian consumers are actively seeking trusted foreign products.
Medical Devices & Diagnostics
CDSCO registration and device classification is a process that most foreign brands underestimate. We manage the full regulatory pathway and connect your product to hospital procurement, diagnostic chains, and clinical distributor networks.
Premium B2B & Industrial
Institutional buyers, including manufacturers, laboratories, and engineering firms, pay reliably and buy repeatedly. We manage B2B sales cycles, technical demonstrations, and key account relationships across India's industrial base.
Specialty Chemicals
Industrial and specialty chemical importers face BIS, MoEFCC, and CPCB compliance requirements on top of standard import processes. We handle the regulatory stack and connect your product to industrial distributors with established client bases.
Why Mittaltiger Market Expansion Services
Not a consultant. Not a distributor. Something different.
Full-stack execution
We don't hand you a report and leave. We build the operation and run it.
Regulatory as a core competency
We hold registrations, manage filings, and maintain compliance so you don't have to hire a regulatory consultant separately.
Asset-light, cost-efficient
No warehouses to fund. No local team to pay. You pay for outcomes, not infrastructure.
Principal-aligned
We only take brands we believe will win in India. Your success is our business model.
Ready to enter India without the complexity?
Let's assess your product and build your market entry plan.